RestoreX® for Lengthening, Post-Prostatectomy
Overall Study
The current study is a randomized, controlled trial of men who underwent prostatectomy. Men are randomized to either receive no treatment (control) or RestoreX for 5 months, beginning one month after prostatectomy. At 6-months following prostatectomy, men are allowed to freely use the device or do nothing for an additional 3 months. The data below are reporting outcomes at the 6-month time point (end of the randomization phase). 82 men have been enrolled in the study. Currently, 6-month results are available on a total of 55 men, with data presented below. These results were published in the Journal of Urology; 206:416-426, August 2021.(1)
Changes in Penile Length
Following prostatectomy, many men report decreases in penile length. Reduced penile length can be associated with worsened hygiene in these men (penile retraction with urinary leakage on surrounding skin) and is a common complaint in sexual medicine clinics. Results from the current study demonstrate significantly longer penile lengths in men who were treated with RestoreX compared to controls.
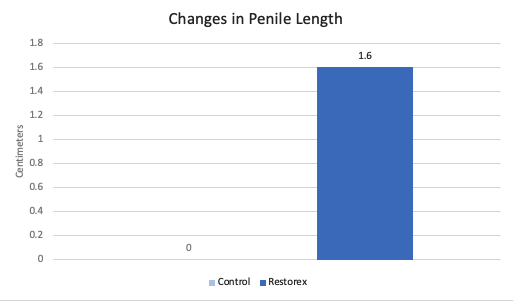
Figure demonstrates significantly longer penile lengths in men treated with RestoreX (p=0.001) compared to controls. The mean change in length for controls was 0 cm compared to 1.60 cm with RestoreX.
Changes in Sexual Function
Following prostatectomy, men most often will experience a worsening of several aspects of sexual function. Results showed that men treated with RestoreX had improved intercourse satisfaction, +1.0, compared to a 3.5 point loss in the control group who had no treatment. Erectile function did not decline in men using RestoreX while the control group ( no treatment ) experienced a 6.5 point decline.
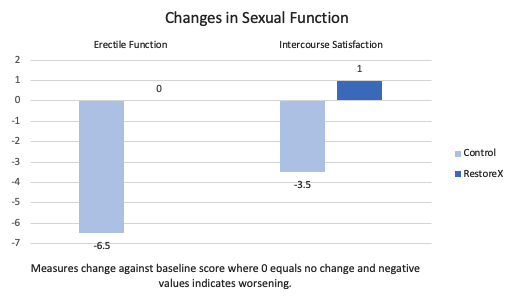
Figure – Results from men who were treated with nothing (control) or RestoreX for 5 months following prostatectomy. Various sexual function domains are represented. Lower = worse outcomes.
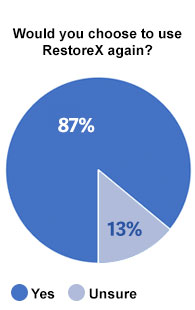
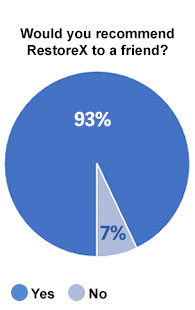
Overall Satisfaction
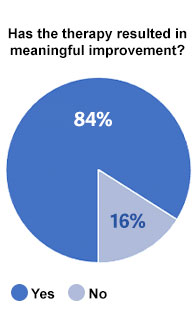
87% of men that were asked if they would choose to use RestoreX again if they were starting the process of recovering from surgery said “Yes.” No men responded “no.” Likewise, 84% of men using RestoreX to recover from surgery said the use of RestoreX was meaningful / worthwhile and 93% would recommend RestoreX to a friend.
- Amir Toussi, Matthew Ziegelmann, David Yang et al. Efficacy of a Novel Penile Traction Device in Improving Penile Length and Erectile Function Post Prostatectomy; Results from a Single-Center Randomized, Controlled Trial; Journal of Urology; 206:416-426, August 2021.
Efficacy of a Novel Penile Traction Device in Improving Penile Length and Erectile Function Post Prostatectomy: Results from a Single-Center Randomized, Controlled Trial
AUA Journals | The Journal of Urology | Urology Practice – August 1, 2021

New device helps restore penile length and sexual function after prostate cancer surgery
Newswise — June 1, 2021 – A new type of penile traction therapy (PTT) device can increase penile length and preserve erectile function in men who have undergone prostate cancer surgery …
